The week of Sept. 21, 2015, saw a flurry of stories about the escalating cost of drugs in the U.S. marketplace. These stories were all initiated by Turing Pharmaceuticals buyout of the drug Daraprim (NY Times Story) and the decision to increase the price per tablet from USD 13.50 to USD 750.00. Turing’s stated reason for the price increase was the fact that the drug is only distributed to about 2,000 users nationally, and therefore at USD 13.50 the drug was not profitable. However, Daraprim is a generic drug that has been on the market for over 70 years, and therefore it is open to competition. In all likelihood, had the price increase held, Turing would have opened itself up to new competition, which would have forced the price back to a more competitive level. In reality, because Turing is currently the only manufacturer, this situation is similar to when a brand drug is coming off patent, and is still able to attract a premium price, before other competitors have time to introduce competing generics and normalize the market.

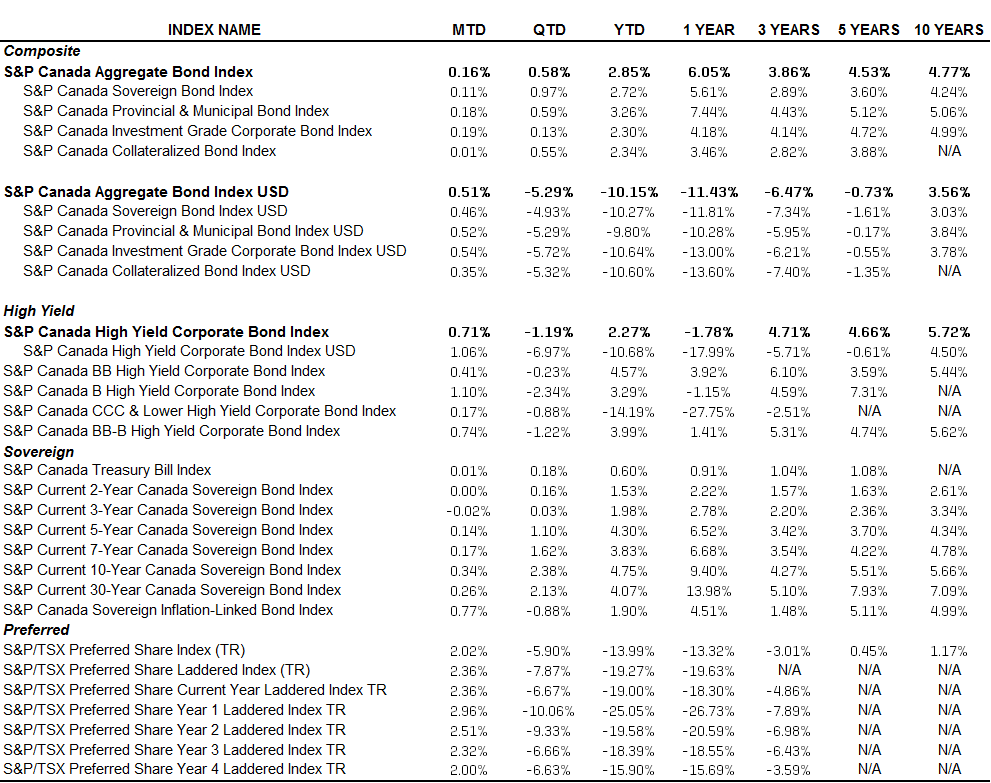
As can be demonstrated by the S&P Healthcare Claims Indices, there are volatile escalations in the overall cost of generic drugs from time to time. These swings are often the result of drugs coming off patent and the resulting market opening for competition. Under the U.S. patent law, drugs coming off patent are open to competition by a single generic producer for a period of six months, and that producer would obviously have a huge price advantage over the market for that period of time. After the initial six month period, the market is open to all generic drug producers to compete. During this phase, we would expect to see costs decrease, as fair market competition brings prices to a market competitive level.
Compared with the overall cost of brand-name drugs, the unit cost of generics is still increasing at a much slower pace. We have seen a significant increase in brand-name drugs recently due to the escalating costs of specialty drugs, such as Sovaldi. With all of that said, in the case of Turing’s drug, a target market of only 2,000 users may not attract huge interest by other manufactures to create a product to compete with, therefore it is possible that a generic drug such as Daraprim could sustain a significantly higher unit cost price than a product with a much wider distribution network. This scenario is based on the facts of competitive markets, and it does not take into account the public reaction that drug manufacturers must also deal with on a daily basis. Anytime a drug manufacturer increases the price of a drug that has a critical role in combating a serious disease by over 5,000%, there is likely to be a public pushback, followed by a political reaction, which may be more severe than the drug manufacturer anticipated.
The posts on this blog are opinions, not advice. Please read our Disclaimers.